What are reduced mitochondria for?
- Dec 4, 2022
- 3 min read
A few years ago, scientists discovered a curious case of protozoa (oxymonads) that do not have mitochondria. Since then, the research group of Associate Professor Hampl from the Faculty of Science of Charles University and the BIOCEV research centre has been asking how the unique loss of mitochondria occurred. In a paper, which has just been published in the prestigious journal Current Biology, Associate Professor Hampl's team with Justyna Zítek as the first author used the closest known relative of the oxymonads, a free living flagellate Paratrimastix pyriformis, as a model species.
This species does have mitochondria, but they are greatly reduced. The team focused on what the closest relative of the mitochondria which has been lost looks like and what this reduced mitochondria actually provides for the organism. In this way, scientists can learn how the reduction of mitochondria proceeded, what makes the oxymonads so unique that they were "lucky" to get rid of the mitochondria altogether.
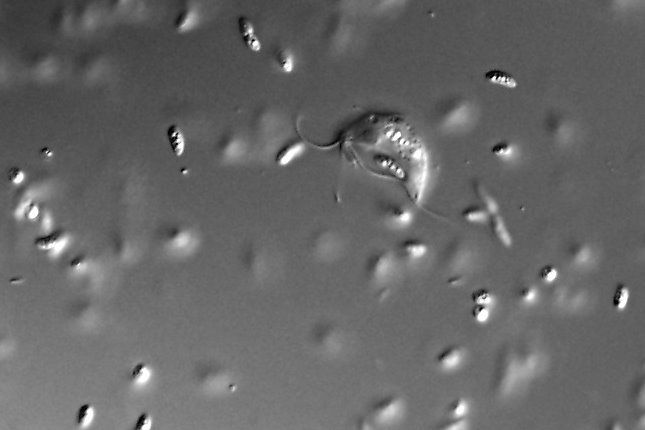
One of the key functions of mitochondria is the synthesis of iron-sulfur clusters, which almost always starts there, even in organisms that have greatly simplified these organelles (e.g. Giardia intestinalis). A few years ago, Hampl's team showed that a whole broader group of oxymonads, which includes the free-living Paratrimastix, has a different pathway for iron-sulfur cluster synthesis that appears to run in the cytosol. "We argued at the time that this pathway exchange is related to the loss of mitochondria in oxymonads. However, we were still interested in how it is in Paratrimastix, because although it has undergone this exchange, it still has mitochondria, so we don't really know how this organelle is beneficial to the cell," says Vladimír Hampl, explaining the background of the new study.

From previous studies, it was clear that the mitochondria of Paratrimastix pyriformis contain a protein complex called the Glycine Cleavage System. This complex is found in a number of other normally free-living anaerobic protists. In the mitochondria of model organisms, including humans, this complex breaks down the amino acid glycine to form NADH, and downstream reactions produce methylated folic acid and formic acid, which are subsequently used in the cytosol for various reactions requiring a methyl group. However, it has not yet been understood what the Glycine Cleavage System is used for in reduced mitochondria.
Justyna Zitek et al. used an advanced proteomic method called LOPIT (Localization of Organelle Proteins by Isotope Tagging), where the cells of an organism are lysed and then several fractions are prepared by differential centrifugation. Researchers prepared nine different pellets - sedimented cell parts. "The cellular components that sediment quickly will be represented more in the first fractions," explains Vladimír Hampl. They then labelled these fractions with isotope tags, mixed them together and measured the mixture in a mass spectrometer. In this way, they identified over five thousand proteins and assigned many of them to cellular compartments. "The trick of the method is that proteins in the same compartment will sediment together and therefore their representation will be similar across fractions, we call it a profile. Because for some proteins we know in advance which compartment they belong to, we know what compartment profile looks like and can match other proteins with it," adds Vladimír Hampl. Using multivariate and advanced classification methods, Justyna Zítek et al. tried to assign proteins to specific compartments in the cell according to their profiles.

The main output of the study is a description of the proteome of the mitochondria of the species Paratrimastix pyriformis, to which statistical analysis has assigned approximately thirty proteins. From this, Zítek et al. were able to deduce that the Glycine Cleavage System is followed by a sequence of three further reactions, resembling the situation in human mitochondria, which generate further folic acid derivatives from the initial product until finally the final product, formic acid, is formed. Vladimír Hampl adds: "It turns out that the reduced mitochondria obviously have an essential function for the organism, which is not the synthesis of iron-sulfur cofactors, but the synthesis of methylated compounds for the cytosol." Nothing similar has been known for simple mitochondria of anaerobic protists.
The work of Justyna Zítek et al. is also key from a methodological point of view, as it shows that the LOPIT technique can be used to study non-model organisms cultured with a mixture of bacteria that are otherwise quite difficult to characterize biochemically or proteomically. "Our study is the second work where the LOPIT method has been applied in full scale to protozoa. It is a very elegant procedure and it was nice to show that it can be applied even to a relatively dirty culture of an organism that we would otherwise find very difficult to study," says Vladimír Hampl.
Provided by Charles University. See original article.



Comments